Case Report - (2021) Volume 8, Issue 5
Massive recurrent penile keloid scar after neonatal circumcision: A case report
Elizabeth Brown*, Amy Ruomei Wu and Matthew D. TimberlakeAbstract
Extensive penile keloid scar formation is a rare complication following newborn circumcision. We present an original case of an extensive recurrent circumferential penile KS in a child managed with surgical excision, z-plasty skin closure, and intralesional triamcinolone injections.
Keywords
Keloid scar, circumcision, pediatric, z-plasty
Introduction
TKeloid Scars (KS) are hyperproliferative growths of collagen in the dermis, often resulting from skin trauma or surgical manipulation. Although benign, KS can result in suboptimal cosmesis, discomfort, and rarely, disfigurement. Pathophysiology involves hyperactive fibroblasts reacting to an extended inflammatory state at the site of injury, resulting in excess collagen deposition locally [1]. Predisposing factors include dark complexion, family history, wound tension, mechanism of injury (e.g. burns), and immune dysfunction [1,2].
Genital KS formation has been infrequently described, and no treatment protocol exists [3,4]. For KS occurring after circumcision, previously-described interventions include excision, steroid injections, radiation therapy, pressure dressings, and use of silicone sheeting. Combination therapy may be the most efficacious in treating and preventing KS formation. We present a case of a massive, disfiguring recurrent penile KS after neonatal circumcision successfully managed with surgical excision, tension-free z-plasty closure and subsequent serial triamcinolone injections.
Case Report
A five year-old Hispanic male presented with a 10 cm × 7 cm × 4 cm circumferential penile mass consistent with a circumcision KS (Fig 1). He had two prior instances of KS formation including a congenital scalp KS that developed due to birth trauma and a ventral penile KS noted at age two. A marginal excision of penile KS was surgically excised at another institution.
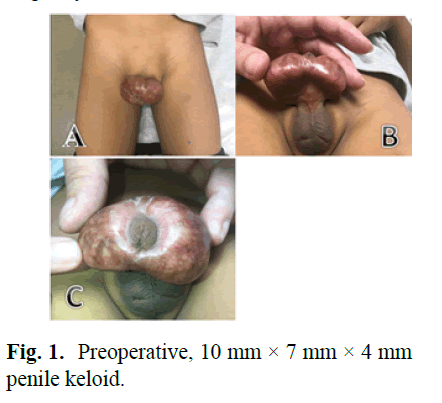
Figure 1: Preoperative, 10 mm × 7 mm × 4 mm penile keloid.
Due to significant disfigurement and poor cosmesis, we performed a marginal excision of the penile KS. A circumferential incision was made along the KS to the level Buck’s fascia and the entirety of the mass was removed using electrocautery and sharp dissection. The specimens were sent for histopathologic analysis. Complex genital skin flaps were then created and a ventral and dorsal midline Z-plasty was performed for skin closure, with great care taken to ensure that there was no anastomotic tension. In order to reduce formation of KS, four rounds of 1.2-1.3 ccs of 40 mg/mL triamcinolone intralesional injections were administered postoperatively along the excision sites at 6-8 week intervals with no recurrence of KS formation (Fig 2).
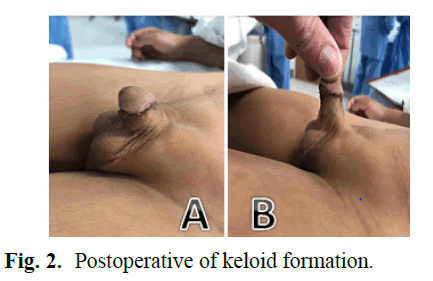
Figure 2: Postoperative of keloid formation.
Discussion
Keloid formation at a cellular level
KS behave as benign, fibrotic scars that expand beyond their original wound boundaries. Studies have shown that KS formation is largely dependent on an autosomal dominant inheritance pattern with incomplete penetration and variable expression and is associated with several different gene polymorphisms associated with apoptosis, inflammation, and the extracellular matrix [5]. KS have also been shown to be more prevalent in darker skinned populations [6]. There have been multiple hypotheses regarding KS formation. Prior studies have shown that lymphocytic infiltration by Th2 lymphocytes drives the fibroblast proliferation resulting in the extensive fibrosis found in KS [2]. It has also been shown that keloid fibroblasts use paracrine signalling to induce surrounding normal fibroblasts and scar fibroblasts to upregulate expression of fibrosis- associated molecular markers and increase cellular activity [1]. Another factor thought to contribute to KS formation is hypoxia causing apoptotic and necrotic regression of vascularity resulting in tissue fibrosis and collagen deposition [2]. There is still significant speculation on the cell-signalling and specific genetic factors that contribute to the formation of KS, but the effect of lymphocytic infiltration, paracrine signaling of keloid fibroblasts, and apoptotic/necrotic pathways induced by hypoxia are thought to result in the proliferating fibrosis characteristic of KS formation.
Keloid formation in response to mechanical stress and the Z-plasty
Our case illustrates the importance of reducing incisional tension during surgical excision to reduce KS formation and recurrence [7]. KS tend to occur more frequently at wound sites with high tensile forces due to dysregulation of mechanotransduction pathways. Mechanosensitive receptors can trigger proliferation of keloid fibroblasts and an abnormal collagen equilibrium, possibly due to disruption of the mechanosignaling pathways that include the TGF-B/Smad, integrin, and calcium ion signalling pathways important to scar formation [8,9]. It has been demonstrated that high tensile forces produce a prolonged inflammation phase in the dermis generating abnormal numbers of blood vessels, collagen, and nerve fibers in the dermal reticular layer. This leads to the endothelial dysfunction found in KS formation [10]. Akaishi concluded from a Finite Element Analysis (FEM) that expansion of KS follow the direction of applied force [11]. Other studies have observed that keloid fibroblasts display significant actin filament stiffness and generate high levels of extracellular matrix when cultured on a stiff substrate [12,13]. These findings suggest that the tension minimizing Z-plasty technique may be most appropriate in patients predisposed to KS formation. The Z-plasty has been shown to release linear scar contractures and tensile forces [14]. The technique alternates vertical and horizontal incisions along the area of KS resection. This is thought to reduce the inflammatory phase of wound healing and promote the proliferative and remodelling phases [15,16]. While the Z-plasty is indicated for scar treatment, there are possible postoperative complications that can arise. With any surgery comes the risk of bleeding, infection, clot formation, and shock. Additionally, the Z-plasty comes with an increased scar length for formation of the flaps and the necessity for skin laxity as well as the possibility of depressed or necrotic skin at acute skin flap angles [16]. There can also be reformation of KS or minimal scar tissue after excision and closure with Z-plasty. Our successful post-operative result suggests that the Z-plasty may be the optimal surgical technique for preventing KS recurrence.
Proposed treatments for penile circumcision keloid
When treating KS, it has been shown that recurrence rates from excision monotherapy near 100%, but when combined with other therapy such as intralesional steroid injections, the rate of recurrence drops to below 50% and 10% with excision and radiation [17]. There has been only one noted case in the literature where excision alone proved successful in KS treatment without recurrence [18]. The use of full thickness skin grafts have also been shown to be useful in the closure after excision of KS with no recurrence [19]. In addition to standard skin graft methods, there have also been some novel skin grafting methods that have had success in treating KS. One study used a novel combination therapy method where the KS borders were incised and irradiated, followed by surgical excision with skin graft closure [20]. There was a statistically significant difference success and satisfaction between the combination therapy group compared to the control group. Radiation therapy has been utilized with great success in the treatment of penile KS, but close proximity of the testes is a limitation of this treatment option [21].
Success has also been demonstrated with the use of a dermal regeneration matrix in addition to epidermal grafting which helps limit tissue contracture and facilitates tension-free wound healing [22]. Local pressure, laser therapy and silicone sheeting have also been used [8,21]. Silicone sheeting is another effective treatment in penile KS, but difficult in application due to location and age of the patient [3,23]. Intralesional steroid injections are another alternative that has been successful in the treatment of penile KS with no recurrence [8,21,24]. Intralesional injections reduce the inflammatory response, thus reducing KS formation. For those genetically predisposed to KS formation, it has been proposed that prophylactic corticosteroid injection be administered to reduce postoperative KS formation [24]. According to the literature, the most popular means of treating penile KS after circumcision has been excision followed by corticosteroid intralesional injections [3,8]. Penile KS post-circumcision have been reported in twenty different cases ranging from 1993 to 2020 with twelve of the patients receiving excision followed by intralesional steroid injections [4,24]. Only one case demonstrated recurrence; subsequently the patient underwent repeat excision with triamcinolone intralesional injections with no recurrence. Four patients underwent intralesional triamcinolone injections only and one with excision only, all without recurrence [18,21,24]. There are also three studies that showed no demonstrated recurrence when intralesional injections were administered prior to surgical excision of the KS [4,22]. Of note, 20 percent of the study population had a prior history of KS formation. Based off the age of our patient and success of surgery along with triamcinolone injections, we chose to pursue this treatment regimen. Although intralesional injections are a recommended first-line treatment for KS, there are possible adverse complications associated with intralesional triamcinolone injections. Local adverse effects include pain with injection or post injection findings such as telangiectasias, changes in pigmentation, skin and subcutaneous fat atrophy and skin ulcerations/ necrosis with risk of local side effects being higher when inadvertently injecting normal tissue [17,25]. Intradermal injection can lead to systemic side effects and toxicity such as Cushing’s syndrome with adrenal insufficiency with a higher predilection occurring in children [25].
Conclusion
Massive recurrent penile KS after neonatal circumcision may be successfully managed with wide excision, tension-free z-plasty closure, then serial local triamcinolone injections. After surgical excision, a combination of treatments includes local mechanical pressure, steroid injection, and silicone sheeting helps to prevent recurrence. However, in order to maintain pressure and silicone film placement, an extra unique dressing procedure is required.
References
- Ashcroft KJ, Syed F, Bayat A. Site-specific keloid fibroblasts alter the behaviour of normal skin and normal scar fibroblasts through paracrine signalling. Plos One. 2013; 8: e75600.
- Appleton I, Brown NJ, Willoughby DA. Apoptosis, necrosis, and proliferation: Possible implications in the etiology of keloids. Am J Patho. 1996; 149: 1441-1447.
- Alyami F, Ferandez N, Koyle MA, et al. Keloid formation after pediatric male genital surgeries: An uncommon and difficult problem to manage. J Pediatr Urol. 2019; 15: 48-e1.
- Buick TA, Abbas W, Munro FD. Literature review and case report of post-circumcision keloid management. Arab J Urol. 2019; 17: 314-7.
- Limandjaja GC, Niessen FB, Scheper RJ, et al. The keloid disorder: Heterogeneity, histopathology, mechanisms and models. Front Cell Dev Biol. 2020; 8: 360.
- Tuan TL, Nichter LS. The molecular basis of keloid and hypertrophic scar formation. Mol Med Today. 1998; 4: 19-24.
- Son D, Harijan A. Overview of surgical scar prevention and management. J Korean Med Sci. 2014; 29: 751-7.
- Mofikoya BO, Adeyemo WL, Abdus-salam AA. Keloid and hypertrophic scars: A review of recent developments in pathogenesis and management. Nig Q J Hosp Med. 2007; 17: 134-139
- Huang C, Akaishi S, Ogawa R. Mechanosignaling pathways in cutaneous scarring Arch Dermatol Res. 2012 ; 304: 589-97.
- Ogawa R, Akaishi S. Endothelial dysfunction may play a key role in keloid and hypertrophic scar pathogenesis–keloids and hypertrophic scars may be vascular disorders. Med Hypotheses. 2016; 96: 51-60.
- Akaishi S, Akimoto M, Ogawa R, et al. The relationship between keloid growth pattern and stretching tension: Visual analysis using the finite element method. Ann Plast Surg. 2008; 60: 445-51.
- Harn HI, Wang YK, Hsu CK, et al. Mechanical coupling of cytoskeletal elasticity and force generation is crucial for understanding the migrating nature of keloid fibroblasts. Exp Dermatol. 2015; 24: 579-84.
- Hsu CK, Lin HH, Harn HI, et al. Caveolin-1 controls hyperresponsiveness to mechanical stimuli and fibrogenesis-associated RUNX2 activation in keloid fibroblasts. J Invest Dermatol. 2018; 138: 208-18.
- Ogawa R. Surgery for scar revision and reduction: From primary closure to flap surgery. Burns Trauma. 2019; 7.
- Hundeshagen G, Zapata-Sirvent R, Goverman J, et al. Tissue rearrangements: The power of the Z-Plasty. Clin Plast Surg. 2017; 44: 805-12.
- Zito PM, Mazzoni T, Z Plasty. StatPearls. Treasure Island. 2020.
- Berman B, Bieley HC. Adjunct therapies to surgical management of keloids. Dermatol Surg. 1996; 22: 126-30.
- Yong M, Afshar K, MacNeily A, et al. Management of pediatric penile keloid. Can Urol Assoc J. 2013; 7: e618-e620.
- Betarbet U, Blalock TW. Keloids: A review of etiology, prevention and treatment. J Clin Aesthet Dermatol. 2020; 13: 33.
- Li W, Wang Y, Wang X, et al. A keloid edge precut, preradiotherapy method in large keloid skin graft treatment. Dermatol Surg. 2014; 40: 52-7.
- Warwick DJ, Dickson WA. Keloid of the penis after circumcision. Postgrad Med J. 1993; 69: 236-237.
- Nguyen KT, Shikowitz L, Kasabian AK, et al. A novel approach to keloid reconstruction with bilaminar dermal substitute and epidermal skin grafting. Plast Reconstr Surg. 2016; 138: 235-9.
- Xie LH, Li SK, Li Q. Combined treatment of penile keloid: A troublesome complication after circumcision. Asian J Androl. 2013; 15: 575-576.
- Hamzan MI, Jeffrey MJ, Halim AS. Rare penile keloid after circumcision: Case report and literature review. Sanamed. 2020; 15: 177-81.
- Coppola MM, Salzillo R, Segreto F, et al. Triamcinolone acetonide intralesional injection for the treatment of keloid scars: Patient selection and perspectives. Clin Cosmet Investig Dermatol. 2018; 11: 387-396.
Author Info
Elizabeth Brown*, Amy Ruomei Wu and Matthew D. TimberlakeReceived: 04-Aug-2021 Accepted: 18-Aug-2021 Published: 25-Aug-2021, DOI: 10.14534/j-pucr.2021267560
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
